Dynamic Sorption Methods
In general, dynamic sorption methods are widely used for characterization of porous materials. With these methods, a gas is flowing over (e.g. dynamic vapor sorption, rapid dynamic flow) or through a sample (e.g. breakthrough curves, inverse gas chromatography). The sample can be weighed and/or the effluent gas flow has to be analyzed. In breakthrough experiments and inverse gas chromatography, the sample has to be arranged as a fixed packed bed. While in breakthrough experiments, a gas flow with an inlet step in gas composition is used, in inverse gas chromatography a pulse function of the composition is applied.
In this article, the breakthrough curve measurement is discussed. For a better understanding the difference between the static volumetric method and a breakthrough experiment will be explained:
Static volumetric measurements:
- Sorption takes place in an enclosed chamber
- Pressure is recorded over time
- Most devices only for determination of single component isotherms
- Measurement of mixtures difficult to carry out, often self-assembled equipment
- hard to realize an automatic setup for measurements of mixtures
Breakthrough Curves (Dynamic Method):
- Sorption takes place in open system
- Pressure is constant
- Outlet composition is recorded over time
- Mixtures only. Measurement of single components hardly possible (only with assumption of non-adsorbable carrier gas) – Therefore: Mixture Adsorption
- Similar to industrial separation process
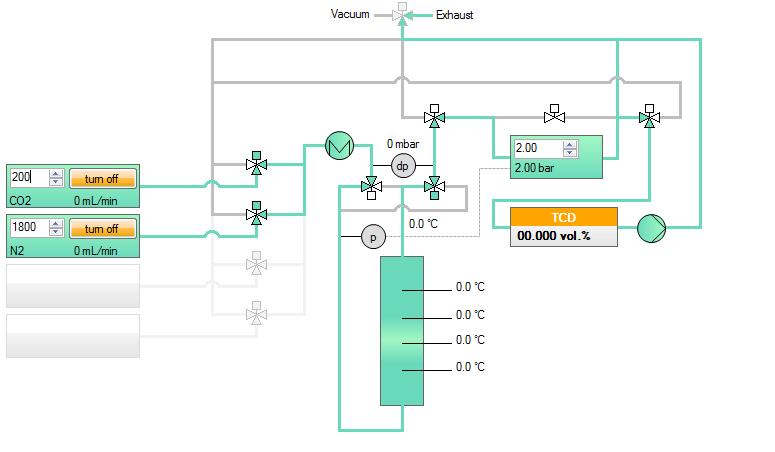
Scheme of an experimental setup
In the next figure a simplified scheme of a breakthrough apparatus is shown. Uusally such a setup consists on a gas dosing and mixing tool (left side in the figure), an adsorber unit (center) and an analytical device or detector, respectively (right side).
Schematic setup to determine a breakthrough curve on a fixed bed adsorber. The shown setup consists of a gas dosing system for a binary mixture, an adsorber with the sample under investigation, and an analytical device to analyze the effluent gas mixture. In this example the adsorber is equipped with 4 temperature probes, a differential pressure sensor and a pressure sensor for absolute pressure in the column.
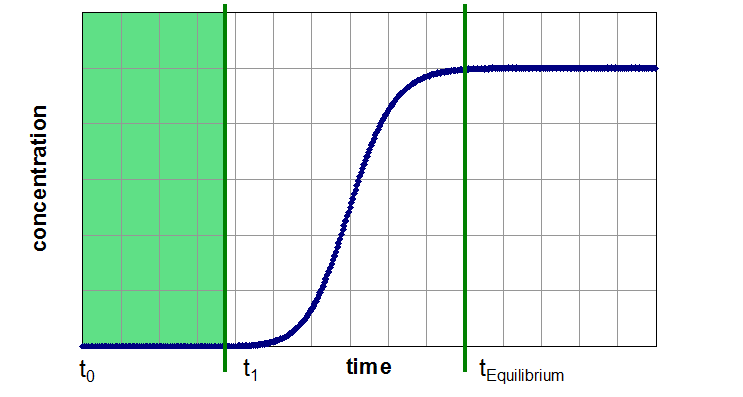
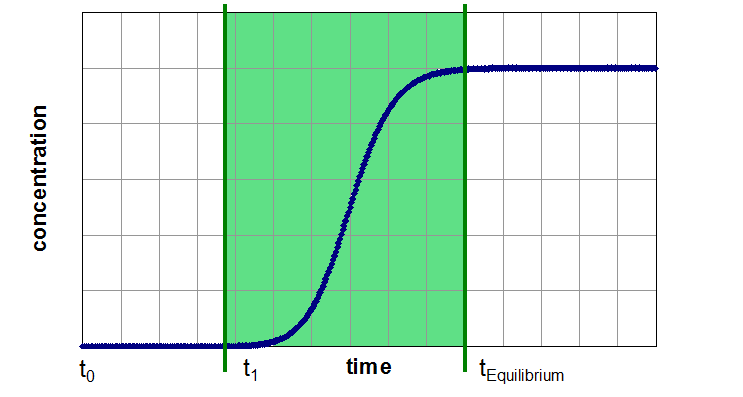
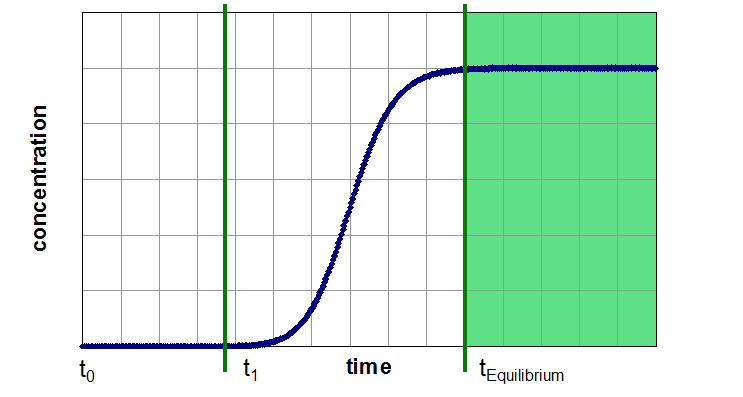
A breakthrough experiment starts typically with switching the inlet gas from a pure carrier gas to a mixture with the desired adsorptive concentration. Therefore, the concentration function at the adsorber inlet is a step function. Now the effluent concentration of the adsorptive is time-resolved measured. During the first part of the experiment, the adsorber can retain the adsorptive a certain time (Segment 1). If the capacity of the adsorbent exceeded, the concentration of adsorptive in effluent gas flow will increase up to the inlet concentration (Segment 2). Afterwards the adsorber is in equilibrium with the gas mixture under the present conditions and no change in gas concentration, temperature or other thermodynamic variables can be observed (Segment 3).
Three Segments of a Breakthrough Curve Experiment
Segment 1: unsaturated adsorber without noticeable breakthrough (production time)
Segment 1 (Unsaturated Zone):
- can be used for determination of a industrially usable sorption capacity
- as a simple benchmark for separation performance of different adsorbens under process relevant conditions (gas velocity, pressure, in presence other adsorptives (e.g. water)
This value can only be determined by dynamic methods
Segment 2: Mass Transfer Zone with rapid increasing of outlet concentration
Segment 2 (Mass Transfer Zone):
- Steepness of curve is influenced by mass transfer, axial dispersion, shape of isotherm, heat effects and heat transfer
- Can be used for qualitative and quantitative (with solving of mass- and energy balances) statements regarding kinetics
- give an insight of the “uneffective” amount of adsorbent during the dynamic process
- Target: Minimize the duration of this zone by optimizing of adsorbent, particle size and process conditions
- With minimized mass transfer zone: Industrially usable sorption capacity very close to saturation capacity
Due to similar conditions like in industrial plants, the kinetic data based on breakthrough curves are in most cases suitable for upscaling.
Segment 3: saturated adsorber, adsorbent is in equilibrium with gas phase
Segment 3 (Saturated Zone):
- Determination of the saturation capacity (similar to equilibrium value, gained from isotherms)
- Measurement of pure component isotherms and mixtures is possible by assuming a thermodynamic controlled system and a non-adsorbable carrier (i.e. He) gas in case of pure component isotherms
- Automated experiments for determination of partial loading over a wide range of partial pressure possible
Due to the possibility of automatization, dynamic sorption methods are one of the most time-saving methods for the determination of mixed gas adsorption equilibria.